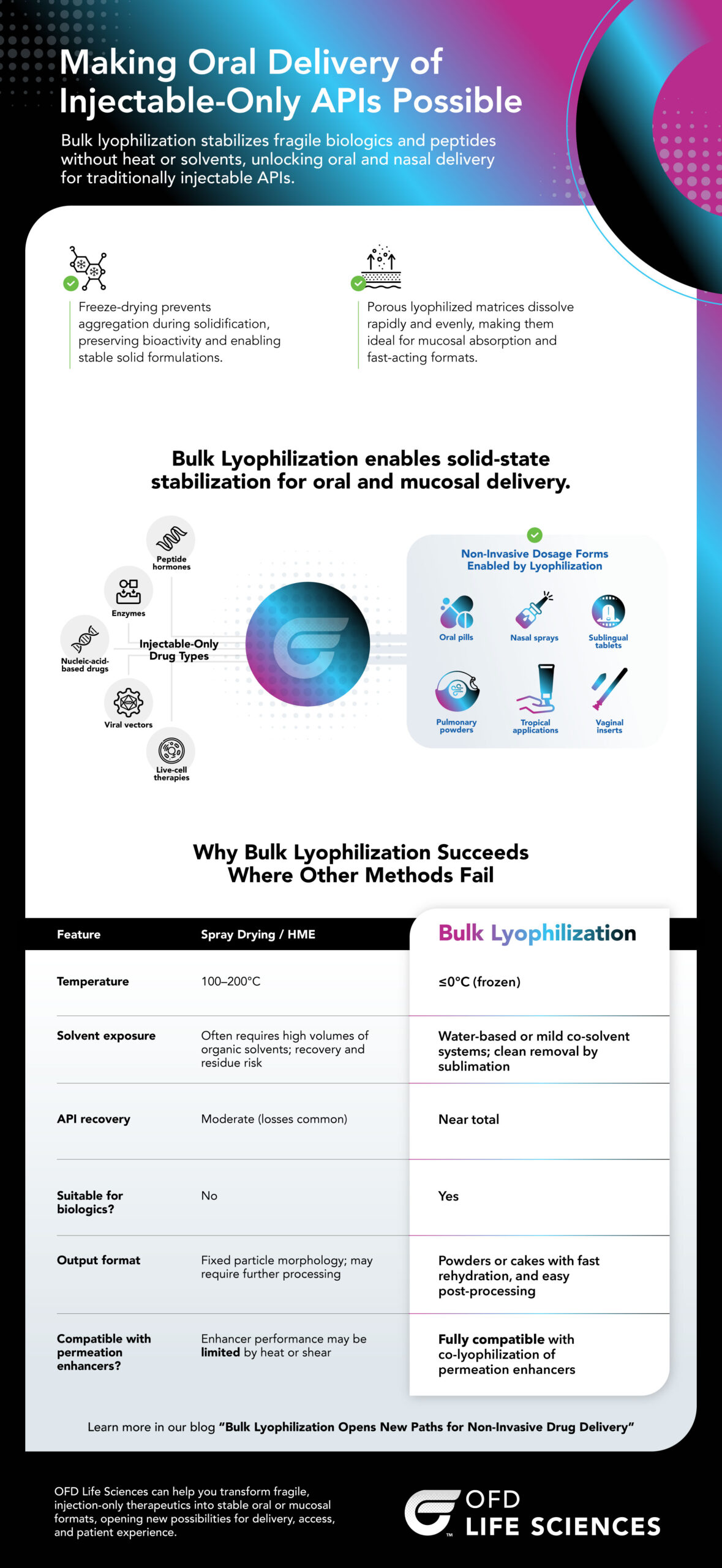
Making Oral Delivery of Injectable-Only APIs Possible
Bulk lyophilization stabilizes fragile biologics and peptides without heat or solvents, unlocking oral and nasal delivery for traditionally injectable APIs.
Freeze-drying prevents aggregation during solidification
Preserving bioactivity and enabling stable solid formulations.
Porous lyophilized matrices dissolve rapidly and evenly
Making them ideal for mucosal absorption and fast-acting formats.
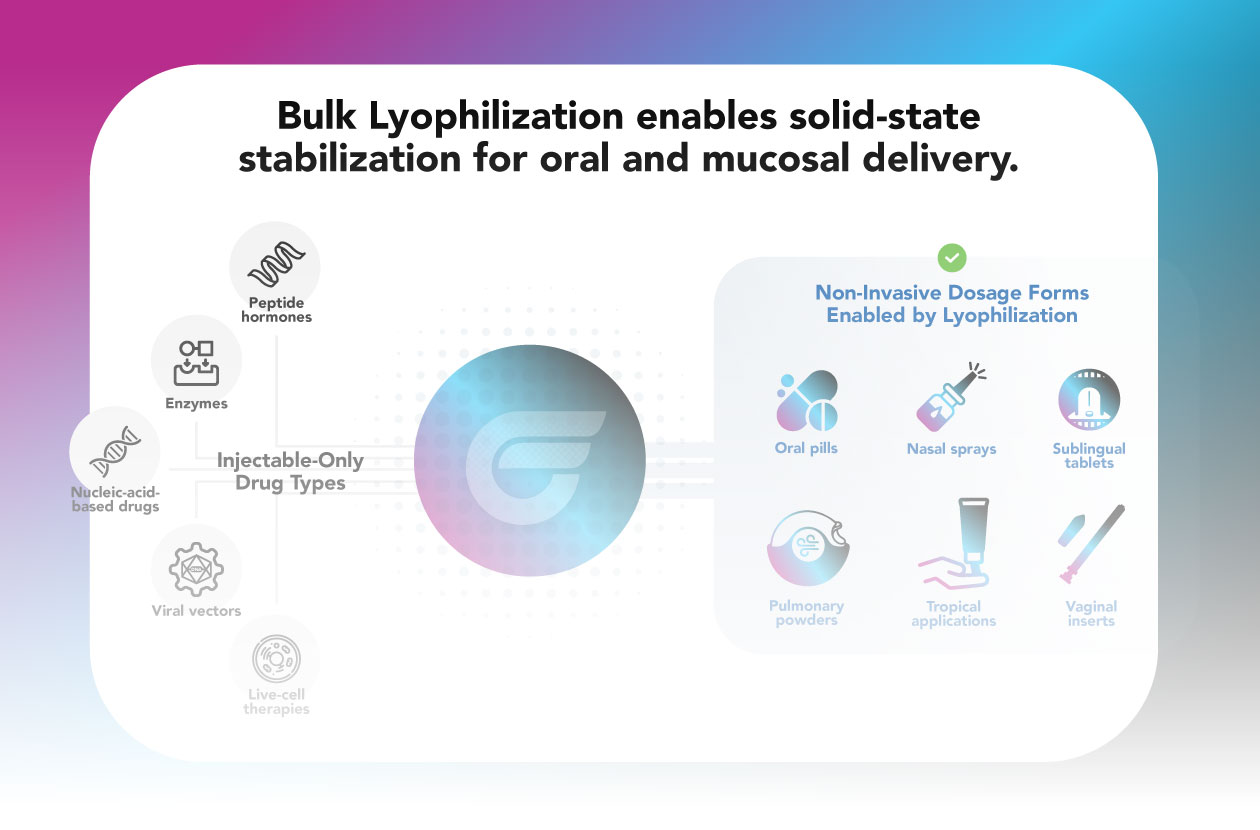
Bulk Lyophilization enables solid-state stabilization for oral and mucosal delivery
Injectable-Only Drug Types
- Peptides
- Enzymes
- Nucleic acid-based drugs
- Viral vectors
- Live-cell therapies
Non-Invasive Dosage Forms Enabled by Lyophilization
- Oral pills
- Nasal sprays
- Buccal tablets
- Pulmonary powders
- Topical applications
- Vaginal inserts
Why Bulk Lyophilization Succeeds Where Other Methods Fail
| Feature | Spray Drying / HME | Bulk Lyophilization |
|---|---|---|
| Temperature | 100–200°C | ≤0°C (frozen) |
| Solvent exposure | Often requires high volumes of organic solvents; recovery and residue risk | Water-based or mild co-solvent systems; clean removal by sublimation |
| API recovery | Moderate (losses common) | Near total |
| Suitable for biologics? | No | Yes |
| Output format | Fixed particle morphology; may require further processing | Powders or cakes with fast rehydration, and easy post-processing |
| Compatible with permeation enhancers? | Enhancer performance may be limited by heat or shear | Fully compatible with co-lyophilization of permeation enhancers |
OFD Life Sciences
OFD Life Sciences can help you transform fragile, injection-only therapeutics into stable oral or mucosal formats, opening new possibilities for delivery, access, and patient experience.
Learn more in our blog “Why Lyophilized Probiotics Stay Alive Longer and Perform Better.”