Drying Methods Comparison
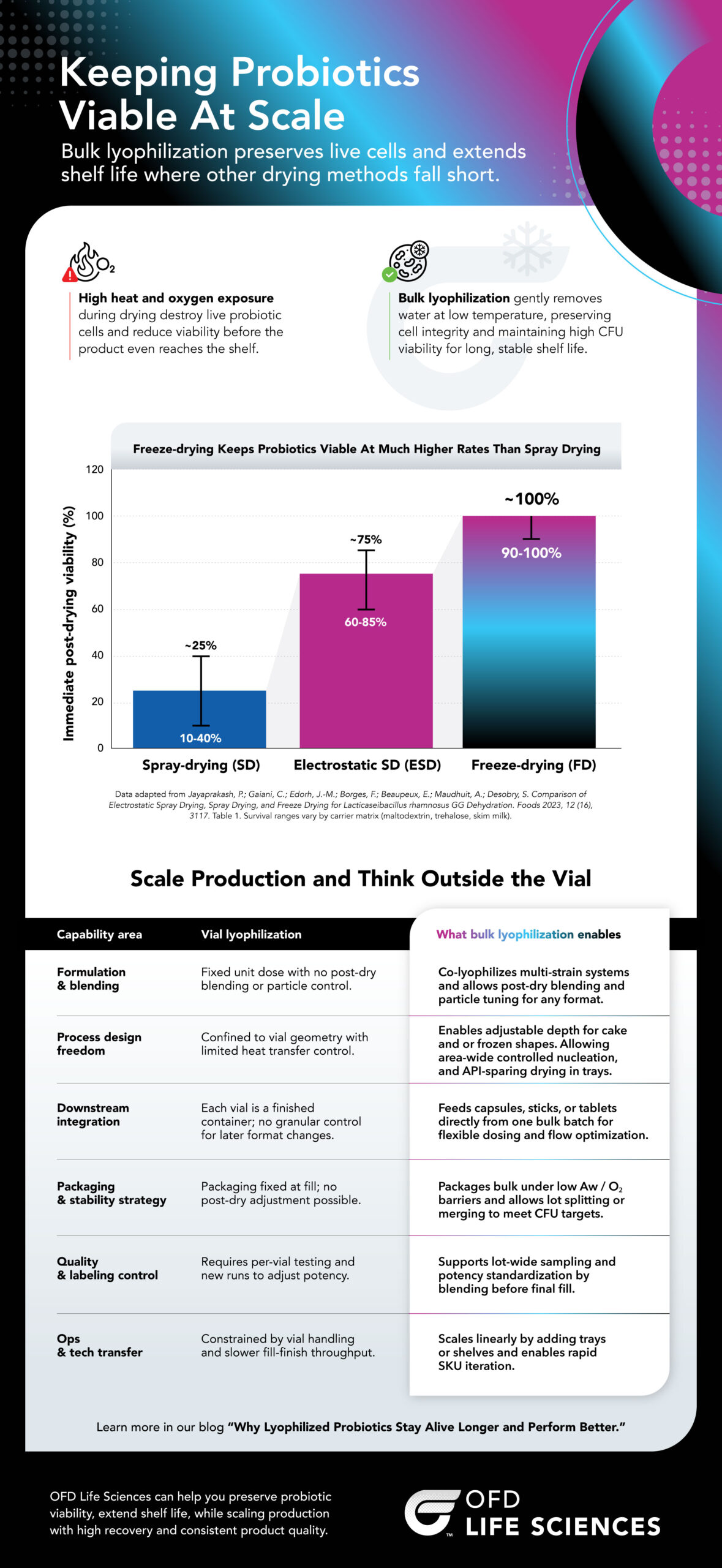
High heat and oxygen exposure
During drying destroys live probiotic cells and reduces viability before the product even reaches the shelf.
Bulk lyophilization
Gently removes water at low temperature, preserving cell integrity and maintaining high CFU viability for long, stable shelf life.
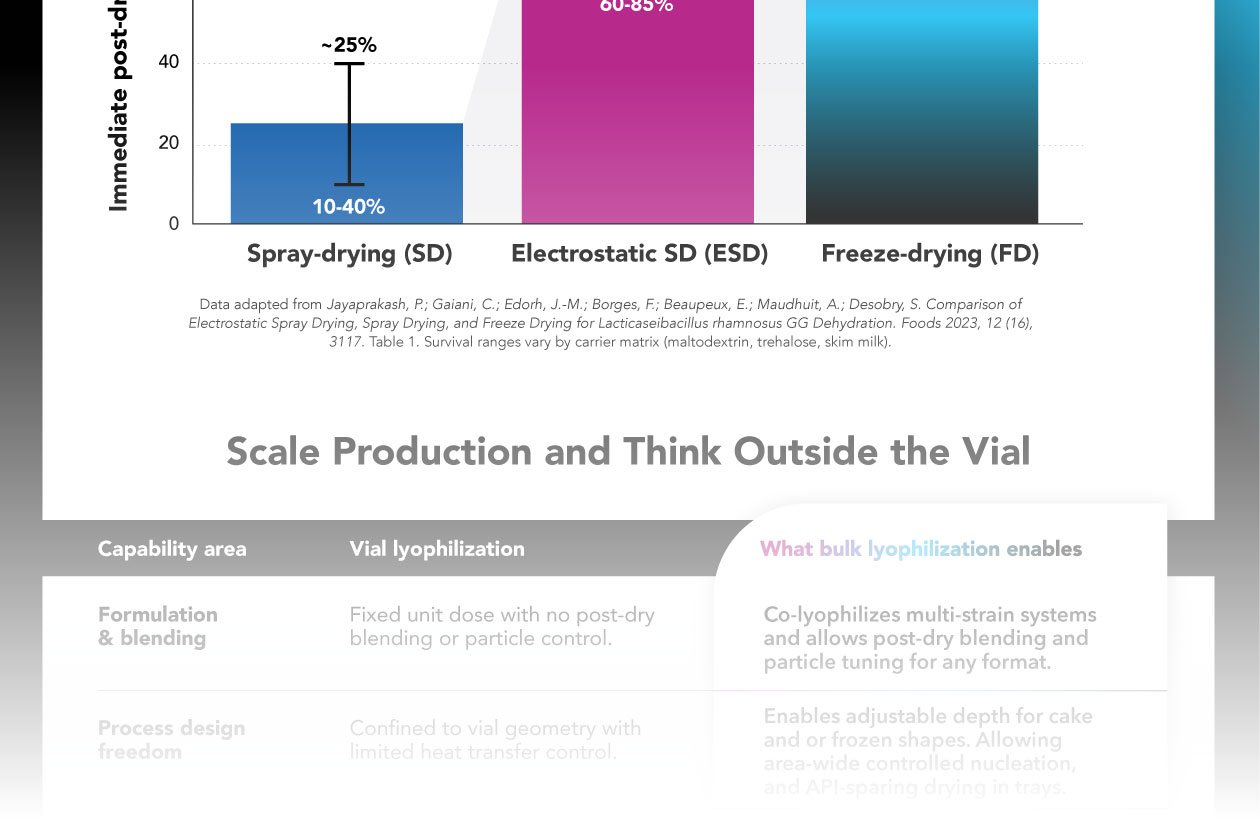
Freeze-drying Keeps Probiotics Viable At Much Higher Rates Than Spray Drying
Immediate post-drying viability (%)
| Drying Method | Average Viability | Viability Range |
|---|---|---|
| Spray-drying (SD) | ~25% | 10-40% |
| Electrostatic SD (ESD) | ~75% | 60-85% |
| Freeze-drying (FD) | ~100% | 90-100% |
Scale Production and Think Outside the Vial
| Capability Area | Vial Lyophilization | Bulk Lyophilization |
|---|---|---|
| Formulation & blending | Fixed unit dose with no post-dry blending or particle control. | Co-lyophilizes multi-strain systems and allows post-dry blending and particle tuning for any format. |
| Process design freedom | Confined to vial geometry with limited heat transfer control. | Enables adjustable depth for cake and or frozen shapes. Allowing area-wide controlled nucleation, and API-sparing drying in trays. |
| Downstream integration | Each vial is a finished container; no granular control for later format changes. | Feeds capsules, sticks, or tablets directly from one bulk batch for flexible dosing and flow optimization. |
| Packaging & stability strategy | Packaging fixed at fill; no post-dry adjustment possible. | Packages bulk under low Aw / O₂ barriers and allows lot splitting or merging to meet CFU targets. |
| Quality & labeling control | Requires per-vial testing and new runs to adjust potency. | Supports lot-wide sampling and potency standardization by blending before final fill. |
| Ops & tech transfer | Constrained by vial handling and slower fill-finish throughput. | Scales linearly by adding trays or shelves and enables rapid SKU iteration. |
Learn more in our blog “Why Lyophilized Probiotics Stay Alive Longer and Perform Better.”